2026.03.23 Patent
Analysis of the Grand Panel Decision of the Intellectual Property High Court (Case No. 2023 (Ne) 10040) ― Manufacturing of Breast Augmentation Composition and Patent Infringement ―

1. Introduction
This article provides an analysis of the Grand Panel Decision of the Intellectual Property High Court (Case No. 2023 (Ne) 10040) rendered on March 19, 2025. This case involves the dispute over whether a physician’s act of preparing a pharmaceutical agent by mixing plasma from blood collected from a person to be treated with commercially available medicines constitutes an infringement of a patent right. This is a significant judicial precedent that makes rulings on legal issues relating to the core of the patent system, such as industrial applicability of an “invention that is a product” relating to medical practices (the main sentence of Article 29, Paragraph (1) of the Patent Act) and the meaning of a “medicine” under the provisions on exemption from liability regarding the act of preparation of a medicine (Article 69, Paragraph (3) of the Patent Act).
2. Description of Technology
The subject patent in this case (Patent No. 5186050) relates to the invention of a composition used for breast augmentation.
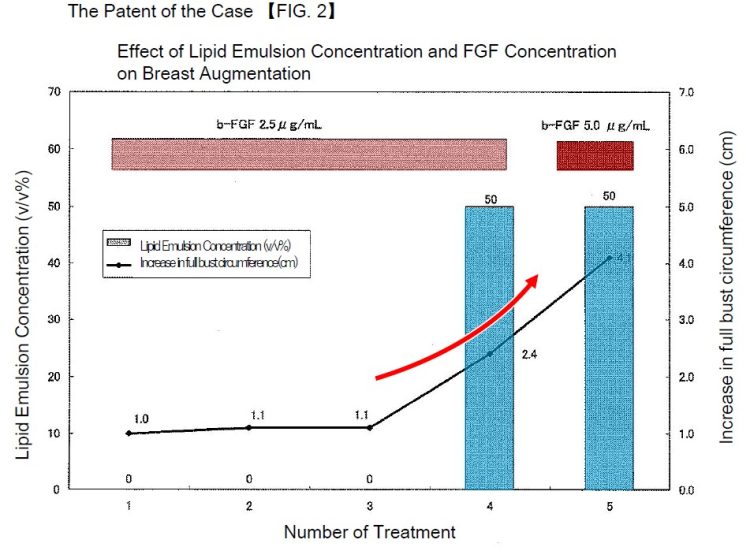
Conventional breast augmentation techniques have included the insertion of implants such as silicone gel bags or the injection of hyaluronic acid. However, these methods have had problems with safety and the durability of effects. The present invention aims to avoid the problems associated with the conventional implant insertion and hyaluronic acid injection, and to generate and increase adipose tissue around the mammary glands to accumulate and increase subcutaneous adipose tissue. Specifically, the subject patent describes that the proteins, lipids, and other constituents contained in the plasma from the person to be treated contribute to the increase of subcutaneous tissue by being combined with the action of b-FGF, and that additionally supplementing a lipid emulsion can effectively promote the accumulation and increase of adipose tissue. The technical feature lies in the combination (inclusion) of the following three components:
|
① Autologous Plasma
A liquid component obtained by centrifuging blood of the person to be treated herself. Among the three components, it is the only component derived from the person to be treated. |
 |
|
② Basic Fibroblast Growth Factor (b-FGF)
A protein that promotes the formation of blood vessels and tissues, specifically, a Trafermin preparation, which is a commercially available medicine. |
 |
|
③ Lipid Emulsion
A commercially available intravenous lipid emulsion for supplementing lipid. The specification of the subject patent exemplifies Intralipid. |
 |
As described above, the subject patent describes that the combination of mixing the plasma derived from the person to be treated with the two commercially available medicines is more effective in accumulating and increasing adipose tissue than using b-FGF or lipid emulsion individually with respect to the plasma.
3. Overview of Case
3-1. Parties and Legal Framework of Case
This is a case where the patentee of the subject patent, Kabushiki Kaisha Tokai Ika (a company engaged in the sale and lease of medical equipment; Appellant / Plaintiff), sued for damages against a physician Y (a cosmetic clinic owner; Appellee / Defendant), claiming that the act of manufacturing a pharmaceutical agent used in the method called “3WAY blood breast augmentation” performed at the Defendant’s clinic constituted an infringement of the patent.
3-2. History from Application to Lawsuit
The patent originates from an application that was filed in 2012 by the physician A as the inventor and was registered in 2013. Kabushiki Kaisha Tokai Ika then received the assignment of the patent right. At the time of the filing, the application included method claims such as “a method for promoting the increase of subcutaneous tissue” and “a method for breast augmentation”. However, these claims were deleted following a reason for rejection stating that they did not constitute “inventions with industrial applicability”. As a result, only the claims relating to an “invention that is a product” (a composition, an injection unit) were registered.
4. Judgment of the Court of First Instance
|
The court of first instance denied the patent infringement at the fact-finding level and dismissed the claim for damages (Tokyo District Court Judgment, March 24, 2023, Case No. 2022 (Wa) 5905).
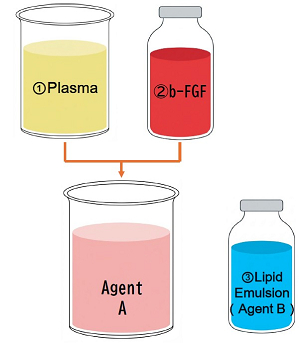
The Plaintiff argued that the Defendant manufactured the composition by mixing the three components (plasma, Trafermin, and Intralipos [a lipid emulsion of the same type as Intralipid, which is exemplified in the specification of the subject patent]).
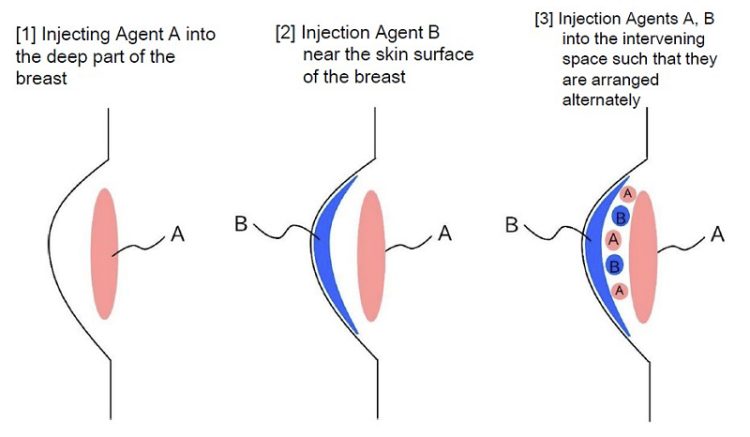
In response, the Defendant countered that he did not mix the components in advance, but instead administered Agent A, which contains plasma and Trafermin, and Agent B, which contains Intralipos, separately into the body.
The court of first instance dismissed the Plaintiff’s claim for damages, finding that there was no evidence to suggest that the Defendant had manufactured the “composition” by mixing these components in advance. The court of first instance did not rule on the other issues (industrial applicability, the exemption provisions under Article 69, Paragraph 3, etc. of the Patent Act).
|
 |
5. Proceedings of the Intellectual Property High Court Grand Panel Case
5-1. Background Leading to the Grand Panel Designation
After the Plaintiff filed an appeal, the Intellectual Property High Court solicited third-party opinions based on Article 105-2-11 of the Patent Act. This system was newly established by the revision of the Patent Act in 2021, and allows the court, at the motion of a party and when deemed necessary, to broadly request the public to submit written opinions regarding the application of laws and other matters relating to the case. Furthermore, the court designated this case as a Grand Panel Case and conducted proceedings.
5-2. Structure of Issues
In the appellate court, in addition to the fact-finding (i.e., whether or not mixing had occurred), legal issues relating to the core of the patent system were delved into deeply, such as the patentability of medical-related inventions and the effect of a patent right on an act of a physician. Among the 11 issues set in this case, the following are particularly important:

5-3. Background to Dispute over the Main Issues
(1) Fact-Finding (Mixed Administration or Separate Administration)
The patent claims describe a composition containing the three components. If the Defendant had administered Agent A and Agent B separately as he claimed, all the three components would not have been present in the syringe prior to administration, thus raising a possibility that his act would not constitute manufacturing the “composition”. Therefore, the actual procedure performed at the site became the primary factual issue.
(2) Industrial Applicability
In Japanese patent practice, “inventions of methods of surgery, therapy, or diagnosis of humans” are not patented as inventions failing to meet the requirements of industrial applicability (JPO Examination Guidelines, Part III, Chapter 1, 3.1.1). However, an invention of a product, such as a medicine, is eligible for patent protection. The point of dispute in this case was whether a “product which uses plasma collected from the body of the person to be treated as part of the ingredients, which is then processed and returned inside the body” is inseparably integrated with medical practices, and thus essentially an “invention that is a process”.
(3) Exemption from Liability regarding Act of Preparation of Medicine (Article 69, Paragraph 3 of the Patent Act)
This paragraph stipulates that a patent right for “a medical invention (medicine meaning a product used in the diagnosis, therapy, treatment, or prevention of human diseases…) that is to be manufactured by mixing two or more medicines” has no effect against the “act of preparation of a medicine as per a physician’s or dentist’s prescription”. In the present case, whether, in the first place, a composition for breast augmentation constitutes the “medicine” (in other words, a product used in … of human diseases) as stipulated in the article became the fundamental question.
Comparison Table of Parties’ Arguments

6. Judgment of the Intellectual Property High Court Grand Panel
The Intellectual Property High Court revoked the judgment in prior instance and found that the Defendant had infringed the patent right. The following explains the judgments for each main issue.
6-1. Fact-Finding: Act of Manufacturing Medicine (Issue 1-2)
Conclusion: The Defendant manufactured (produced) a composition in which the three components were mixed.
The court comprehensively evaluated the descriptions in the medication notes made at the clinic, the descriptions in the documents provided to the persons to be treated, and the descriptions in the advertisements on the website, and determined that the Appellee prepared a pharmaceutical agent simultaneously containing the three components and administered it to the person to be treated. As a result, it became unnecessary to determine whether the case in which the components are administered separately and then mixed inside the body would constitute the “production” (Issue 1-3).
6-2. Industrial Applicability (Issue 2-1)
Conclusion: The subject invention constitutes the “invention with industrial applicability” (valid patent).
First, the court pointed out that the main sentence of Article 29, paragraph (1), does not explicitly exclude products administered to the human body from the subject matter of patent, and the background of the 1975 revision where the former Article 32, item (ii) of the Act prior to that amendment, which had specified medical inventions non-patentable, was deleted. Based on the above, the court stated that it is difficult to construe that an “invention that is a product” is substantially an “invention that is a process” and that it does not constitute an “invention with industrial applicability”, solely because the pharmaceutical agent is intended to be administered to the human body.
Regarding the use of autologous plasma, the court also held that an act of manufacturing a medicine or the like by using an ingredient collected from humans is not necessarily performed only by a physician, and that blood collection, manufacturing the composition, and its administration are not always a series of acts that are integrated into one. The court concluded that development of advanced medical technologies such as regenerative medicine greatly owes to research and development in the pharmaceutical industry, etc., and there is a necessity to grant patent protection to promote technical development.
6-3. Exemption from Liability regarding Act of Preparation of Medicine (Issue 3-2)
Conclusion: The subject composition does not constitute the “medicine” in Article 69, Paragraph 3, and the exemption provision is not applicable.
The court held that the purpose of the subject composition is mainly aesthetic, because the claims include an explicit recitation “a composition (…) used for breast augmentation”, and the specification includes a description “(…) for the purpose of achieving a beautiful appearance”. Based on the above understanding and in light of the general meaning of “disease” (“a phenomenon in which an organism suffers an abnormality in the physiological state of the whole or part of its body, an inability of the body to perform normal function, and various types of pains” (c.f., the Kojien, Japanese dictionary, etc.), the court determined that it is difficult to say a state requiring a breast augmentation operation, which is mainly for aesthetic purposes, is a “disease”, and it is also difficult to recognize that the subject composition is a “product used in the… therapy, treatment or prevention of human diseases…”.
In addition, regarding the legislative intent of Article 69, Paragraph 3, the court stated that the gist of this provision is that the preparation of a medicine is conducted by way of a physician selecting a medicine that is expected to have the most appropriate efficacy for therapy, etc. of a human disease and preparing a medicine as per a prescription, and it should be ensured that the selection by the physician would not be obstructed by a patent right, from the viewpoint of “realizing the public interest of smooth implementation of medical practice”; however, such public interest cannot be immediately found at least for the selection of the pharmaceutical agent to be used for breast augmentation according to the subject invention.
Therefore, without further consideration of whether the Defendant’s act constitutes “an act of preparation of a medicine as per a prescription”, the application of Article 69, Paragraph 3 was denied at the stage of determining whether the composition qualifies as a “medicine”.

7. Significance of the Judgment and Implications for Practice
7-1. Affirmation of Patentability and Enforcement for Product Inventions Containing Human-Derived Raw Materials
Conventionally, technologies that involve “processing materials collected from patients and returning them into the body”, such as those used in customized medicine and regenerative medicine, have had ambiguous boundaries with medical practices, and have been considered difficult to enforce. This judgment clearly stated that an “invention that is a product which uses an ingredient collected from a human and which is intended to be finally returned inside the body of that human” is also industrially applicable and subject to patent protection. This decision can be considered as the court’s straightforward endorsement of the Japan Patent Office’s practice of handling a medicine as an invention of a product and made it patentable, and it holds great significance as a practical guideline for filing a patent application for an invention of a product containing a human-derived ingredient in the bio-healthcare field.
7-2. Judgment on Scope of “Medicine” in Article 69, Paragraph 3
It is of great significance that the court issued an unambiguous judgment on the definition of “medicines” in Article 69, Paragraph 3 of the Patent Act. The breast augmentation composition of the present invention was deemed not to constitute a “product used in…therapy… of human diseases”, and the exemption on the ground of the act being a physician’s act of preparing a medicine was denied. This determination indicates that the exemption under Article 69, Paragraph 3 does not extend at least to the preparation of a medicine mainly for aesthetic purposes. However, the judgment only ruled on the breast augmentation composition, and the court did not provide a determination on the application of Article 69, Paragraph 3 in areas where, for example, therapeutic and aesthetic purposes are inseparably connected. We will wait and see what standard the court will present in such borderline cases in the future.
7-3. Issue 1-3 (Whether Separate Administration Constitutes “Production”) Remains Unresolved
Judgment on the issue “whether a case where the three components are administered separately and mixed inside the body constitutes the ‘production'”, which was one of the most anticipated issues in this case, was avoided as a result of the fact-finding. This is an extremely important practical issue as it concerns whether patent infringement can be avoided by devising administration methods in the medical field, and it may be disputed again in future cases.
7-4. Practical Lessons on Establishing Infringement
When proving infringement, the point that the manufacturing act (mixing) was inferred from the descriptions in the “medication notes” used at the site, the consent forms, and the website advertisements is an important guideline for a practice of collecting evidence of medical practices performed in a closed environment. For patentees, it is essential to continuously collect and preserve evidence that can lead to the inference of the contents of suspected infringing acts.
Edited by Kenshi Takenaka

